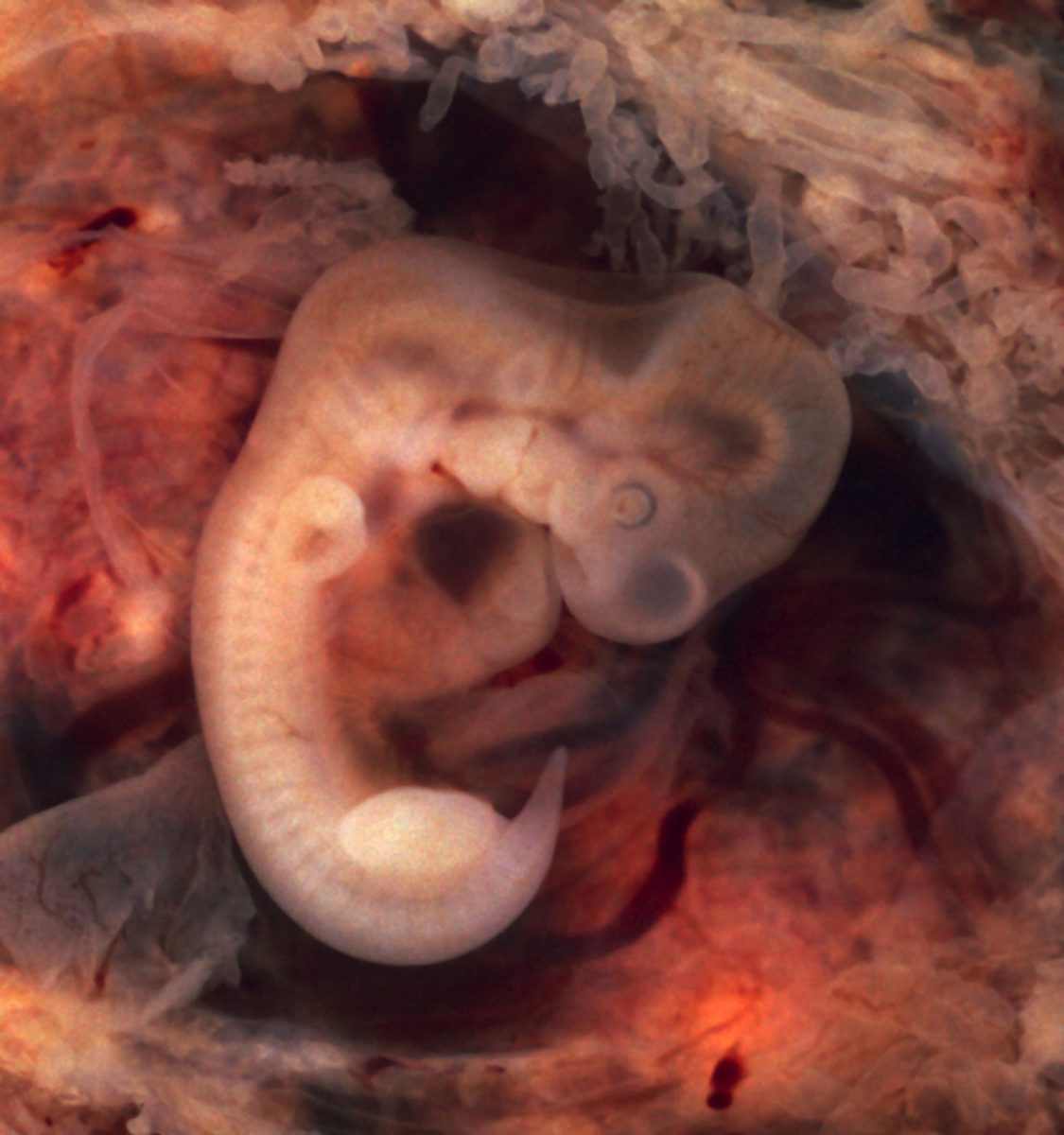
About one in 10 infants in the Unites States are born premature. These infants need special around-the- clock medical care until their organ systems can work independently, and yet they still usually have long-term health problems. What if there were a way for a premature infant to live in a womb after birth so he or she can develop properly?
Researchers at the Children’s Hospital of Philadelphia Research Institute have made major steps in creating a working artificial womb. Premature fetal lambs, being similar to premature human infants, were able to develop in the artificial womb for up to four weeks. Their system is different from past artificial womb systems, as it allows gas exchange to the fetus in a “closed, sterile, fluid environment,” as reported in Nature Communications in April.
When human infants are supported by gas-based ventilation systems in the neonatal intensive care unit, as opposed to liquid-based ventilation in the womb, their lungs do not develop as a healthy infant’s would. This leads to a long-term decrease of function and lifelong morbidities. However, the eight lambs that survived in the device were connected to machinery via their umbilical cords. This connection allowed fluid gas exchange directly to their developing lungs. Once the lambs were birthed, their lung function was “essentially caught up” to the lung function of mature lamb infants.
The system aims to replace the function of the placenta and mimic the environment of the womb. Like the placenta, there is gas exchange occurring through the umbilical cord. The connection allows blood to flow from the fetus to an oxygenator, a machine which oxygenates blood, and back to the fetus. Like the womb, the environment surrounding the fetus is fluid, allowing the fetus to swallow and breathe amniotic fluid.
The research team hopes to begin testing with humans soon. They are currently working with the FDA to design a preclinical study to test the safety and efficacy of the system more in depth.
Although the treatment will not be used in the neonatal ICU any time soon, this study is a major development toward a solution for premature births.
Categories:
Tigra Scientifica: Womb for Improvement
Grace McNamara, Contributor
October 30, 2017
0
Donate to The Tiger
Your donation will support the student journalists of Clemson University. Your contribution will allow us to purchase equipment and cover our annual website hosting costs.
More to Discover